The advent of biologic therapy has made it possible to set new standards of efficacy and safety in the treatment of moderate to severe psoriasis, which are far reaching and uniformly recognized on the basis of evidence from clinical trials, registries and meta-analyses. Economic considerations are the main reason why some guidelines and most of the agencies responsible for policies on the reimbursement of prescriptions in the European Union (EU) and Spain position biologic drugs as second-line treatments and limit their use to patients who present intolerance to classic systemic treatments (methotrexate, acitretin, ciclosporin), and phototherapy or cases in which these therapies have failed or are contraindicated.1 From a strictly evidence-based perspective, the expectation of efficacy is generally lower for the conventional systemic treatments than for all of the biologic therapies (with the possible exception of etanercept, a biologic drug now rarely considered as an option in dermatology); moreover the conventional agents are associated with a higher risk of drug interactions and organic toxicity and, overall, have a worse safety profile.2 If economic considerations were not the primary concern, the biologics would be first-line therapies and the conventional systemic agents would be positioned as second-line treatments.
In many European countries, the introduction of biosimilars into the health system has radically changed prescription patterns and increased the involvement of hospital pharmacists and health care payers as significant stakeholders in the decision-making process. The impact and eventual repercussions of this change on the clinical management of patients with moderate to severe psoriasis are not yet clear. While decreases in the acquisition cost of biologic agents may reduce the economic burden associated with the choice of certain drugs to treat psoriasis and thereby facilitate broader access to such therapies, the new situation has also given rise to other developments that are, or may prove to be, negative. While the efficiency of any treatment can be evaluated on the basis of objective data (for example, the cost per patient of achieving a 90% or greater improvement in Psoriasis Area Severity Index [PASI-90] as compared to baseline),3 decisions are very often based solely on the “price” factor rather than on measures of efficiency. The introduction of biosimilars appears to be influencing institutional decisions (Treatment Appraisal Reports) on the reimbursement criteria for some of the drugs most recently approved for the treatment of psoriasis. Unwarranted restrictions affecting the reimbursement of new original molecules have been introduced. These new rules are unrelated to the previous Treatment Appraisal Reports and place class criteria (tumor necrosis factor [TNF] inhibitors) before efficiency (available biosimilars on the basis of cost per patient achieving an adequate response). The new regulations lead to inequitable access and complicate the decisions of patients and physicians in clinical practice.4
Definition of Biosimilarity Based on the Totality-of-the-Evidence ApproachBiosimilars are biologic medical products highly similar to other already approved biologic agents (the originator or reference biologics) without clinically meaningful differences in terms of safety, purity, and potency relative to the reference products, although they may differ in terms of the manufacturing process and the excipient or delivery device used.5 Amino acid sequences can (and must) be reproduced exactly, but in a complex manufacturing process, such as that of biologic agents, multiple factors can be introduced that may result in alterations in the protein structures or post-translational modifications (e.g. in glycosylation) that can affect the efficacy, immunogenicity and/or safety of the final product. For this reason, regulatory agencies require complete preclinical analytical studies of biosimilars including over 200 determinations, complemented by pharmacokinetic/pharmacodynamic studies in healthy volunteers or patients, and at least 1 clinical study confirming the therapeutic equivalence of the biosimilar and the reference biologic in a patient population chosen to maximize statistical sensitivity.6,7
Minimal differences in inactive components are permitted provided they do not affect the safety, purity and potency of the drug. By contrast, variations between batches of the reference biologic do occur (due to changes in the process or the factory), in some cases giving rise to greater variability in the potency of the reference product than is found in the biosimilar.8 Although such alterations or differences between batches do not usually affect the quality of the product,9,10 recently published evidence has indicated that they may have implications for the therapeutic efficacy of trastuzumab.11,12 In the case of biosimilar monoclonal TNF-α inhibitors, 2 aspects should be evaluated: TNF-α binding capacity and Fc domain-mediated antibody effector functions, such as complement-dependent cytotoxicity and antibody-dependent cell-mediated cytotoxicity.
The biosimilarity of the proposed biosimilar is also assessed in a pharmacokinetic study using a single-blind parallel group or double-blind crossover design in which healthy individuals or patients receive a predetermined number of doses of the drug intravenously or subcutaneously and blood samples are obtained for pharmacokinetic study (maximum concentration, half-life, and area under the concentration-time curve) and to determine immunogenicity (anti-drug and neutralizing antibody titers). Drug safety is also assessed.
The final step is to establish that the efficacy and safety of the biosimilar is similar to that of the reference biologic, often by way of an equivalence trial; the margin of error used in biosimilar studies in psoriasis ranges from ±14% to ±18%.13 The primary endpoint of the study and the time of the determination may or may not be the same as those used in the pivotal trials of the reference biologic, but the number of patients included is generally lower due to the characteristics of the statistical design, which are currently the subject of active research.14,15 The smaller sample size and the fact that studies for each indication are not required, provided the criteria for interchangeability are met, means that the cost of the clinical development of biosimilars is lower than that of “innovative” biologics. In fact, as our experience in the preclinical analytical comparison process increases and the equivalence of biosimilars introduced into clinical practice is confirmed, the need to include therapeutic equivalence trials for TNF-α inhibitors is being discussed.16 It is recommended that the clinical trial should, at least, be undertaken for the indication that includes a more sensitive population in order to detect possible differences (for example, psoriasis, given the low rate of placebo response and the absence of any concomitant medication that might reduce the immunogenicity of the originator biologic and the biosimilar).7 The following anti-TNF-α biosimilars approved by the European Medicines Agency for the same indications as their corresponding reference biologic agents have been tested for equivalence in psoriasis: Erelzi® (Sandoz), Amgevita® (Amgen), Hyrimoz®/Halimatoz®/Hefiya® (Sandoz), Cyltezo® (Boehringer Ingelheim), and Idacio®/Kromeya® (Fresenius Kabi).17 Of the other biosimilars listed in Table 1, the following have been tested in patients with psoriasis: BCD-057, M923, MYL-1401A, CHS-1420, and ONS-3010 (search www.clinicaltrials.gov and www.clinicaltrialsregister.eu).
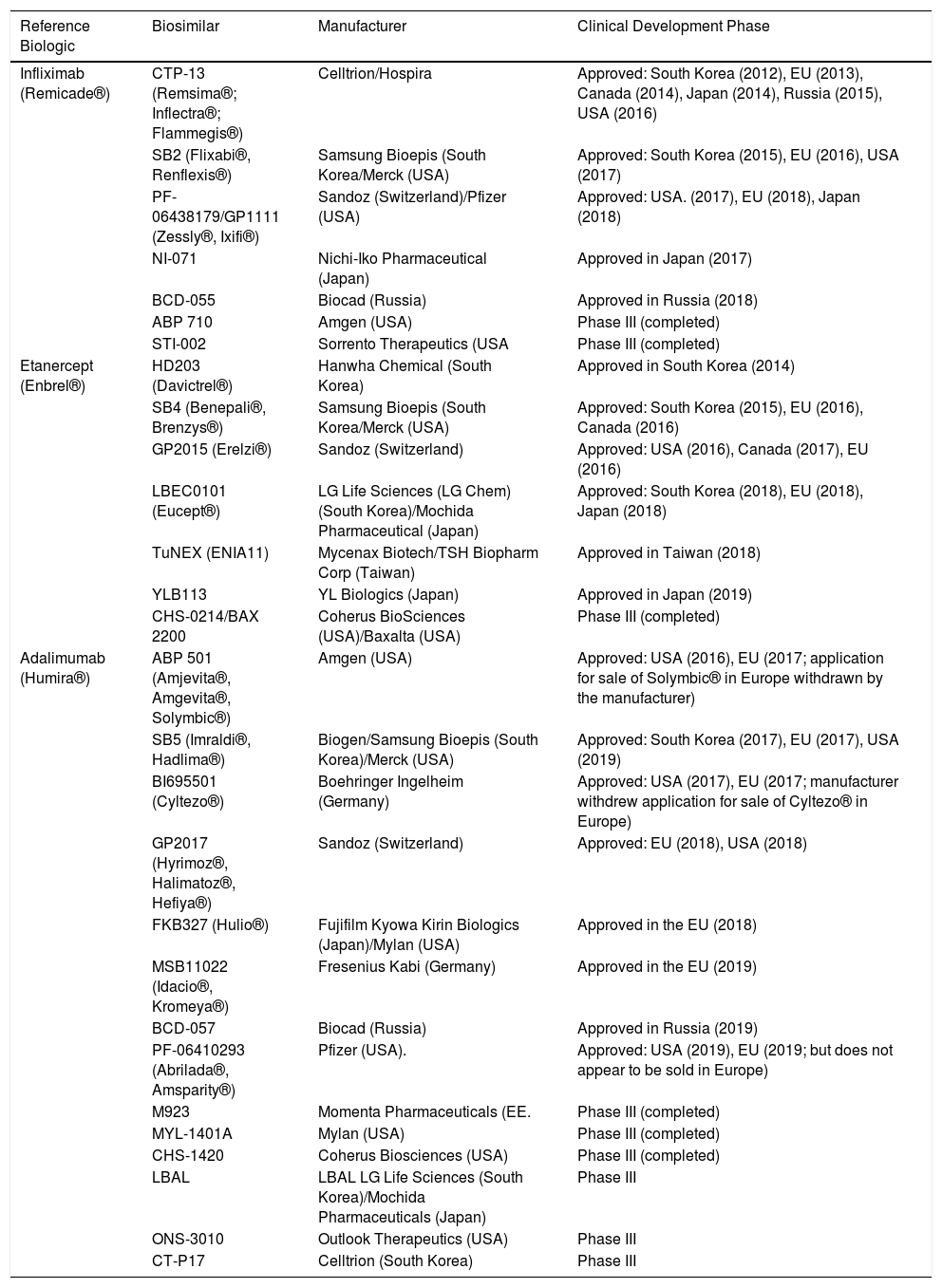
Tumor Necrosis Factor α (TNF-α) Inhibitor Biosimilars Which Are Approved or in Phase III of Clinical Development.
| Reference Biologic | Biosimilar | Manufacturer | Clinical Development Phase |
|---|---|---|---|
| Infliximab (Remicade®) | CTP-13 (Remsima®; Inflectra®; Flammegis®) | Celltrion/Hospira | Approved: South Korea (2012), EU (2013), Canada (2014), Japan (2014), Russia (2015), USA (2016) |
| SB2 (Flixabi®, Renflexis®) | Samsung Bioepis (South Korea/Merck (USA) | Approved: South Korea (2015), EU (2016), USA (2017) | |
| PF-06438179/GP1111 (Zessly®, Ixifi®) | Sandoz (Switzerland)/Pfizer (USA) | Approved: USA. (2017), EU (2018), Japan (2018) | |
| NI-071 | Nichi-Iko Pharmaceutical (Japan) | Approved in Japan (2017) | |
| BCD-055 | Biocad (Russia) | Approved in Russia (2018) | |
| ABP 710 | Amgen (USA) | Phase III (completed) | |
| STI-002 | Sorrento Therapeutics (USA | Phase III (completed) | |
| Etanercept (Enbrel®) | HD203 (Davictrel®) | Hanwha Chemical (South Korea) | Approved in South Korea (2014) |
| SB4 (Benepali®, Brenzys®) | Samsung Bioepis (South Korea/Merck (USA) | Approved: South Korea (2015), EU (2016), Canada (2016) | |
| GP2015 (Erelzi®) | Sandoz (Switzerland) | Approved: USA (2016), Canada (2017), EU (2016) | |
| LBEC0101 (Eucept®) | LG Life Sciences (LG Chem) (South Korea)/Mochida Pharmaceutical (Japan) | Approved: South Korea (2018), EU (2018), Japan (2018) | |
| TuNEX (ENIA11) | Mycenax Biotech/TSH Biopharm Corp (Taiwan) | Approved in Taiwan (2018) | |
| YLB113 | YL Biologics (Japan) | Approved in Japan (2019) | |
| CHS-0214/BAX 2200 | Coherus BioSciences (USA)/Baxalta (USA) | Phase III (completed) | |
| Adalimumab (Humira®) | ABP 501 (Amjevita®, Amgevita®, Solymbic®) | Amgen (USA) | Approved: USA (2016), EU (2017; application for sale of Solymbic® in Europe withdrawn by the manufacturer) |
| SB5 (Imraldi®, Hadlima®) | Biogen/Samsung Bioepis (South Korea)/Merck (USA) | Approved: South Korea (2017), EU (2017), USA (2019) | |
| BI695501 (Cyltezo®) | Boehringer Ingelheim (Germany) | Approved: USA (2017), EU (2017; manufacturer withdrew application for sale of Cyltezo® in Europe) | |
| GP2017 (Hyrimoz®, Halimatoz®, Hefiya®) | Sandoz (Switzerland) | Approved: EU (2018), USA (2018) | |
| FKB327 (Hulio®) | Fujifilm Kyowa Kirin Biologics (Japan)/Mylan (USA) | Approved in the EU (2018) | |
| MSB11022 (Idacio®, Kromeya®) | Fresenius Kabi (Germany) | Approved in the EU (2019) | |
| BCD-057 | Biocad (Russia) | Approved in Russia (2019) | |
| PF-06410293 (Abrilada®, Amsparity®) | Pfizer (USA). | Approved: USA (2019), EU (2019; but does not appear to be sold in Europe) | |
| M923 | Momenta Pharmaceuticals (EE. | Phase III (completed) | |
| MYL-1401A | Mylan (USA) | Phase III (completed) | |
| CHS-1420 | Coherus Biosciences (USA) | Phase III (completed) | |
| LBAL | LBAL LG Life Sciences (South Korea)/Mochida Pharmaceuticals (Japan) | Phase III | |
| ONS-3010 | Outlook Therapeutics (USA) | Phase III | |
| CT-P17 | Celltrion (South Korea) | Phase III |
It is interesting to note that in many equivalence studies in patients with psoriasis or rheumatoid arthritis, biosimilars obtain response rates higher than those reported in the pivotal studies for the originator biologic product; this may be due to the use of a study design without a placebo arm, which would tend to raise researchers’ expectations with regard to efficacy.18
In terms of safety and immunogenicity, the results of the clinical trials confirm the biosimilarity within the margin of equivalence, although variations in the excipients used may justify some small differences. For instance, Erelzi® causes fewer injection site reactions than Enbrel®.19 In the case of adalimumab biosimilars, the reference drug used in equivalence trials has always been the formulation of Humira® manufactured before the changes introduced to reduce injection pain (elimination of citrate from the vehicle, the use of a smaller volume, and delivery with a smaller gauge needle).20 Of the biosimilars currently available, only Hulio® does not contain citrate or acetate in its excipient.21
To meet the requirements of the US Food and Drug Administration for demonstrating biosimilar interchangeability, most clinical trials of biosimilar TNF-α inhibitors include an extension study involving a single switch from originator to biosimilar or a crossover design with randomization and one or more switches between the two products.22 In the case of Hyrimoz®/Halimatoz®/Hefiya®, the results of a study with multiple switches in patients with psoriasis are available.23 Although the size and duration of these studies may not be sufficient to detect possible differences, there is scientific evidence of comparability between 2 biosimilars supporting the efficacy and safety of interchanges, even between biosimilars.24 This could, in the medium term, mean that biosimilars with the same active ingredient could be interchanged without any consideration of their technical differences and solely on the basis of variations in local or national financial agreements. In such a scenario, the clinician’s role in the decision making process may be greatly reduced except when treatment with a biosimilar (or originator biologic product) fails or is contraindicated. It is, nevertheless, important for scientific societies and opinion leaders to bear in mind that in this setting, as in any other area of medical therapeutics, clinicians must play a central role in decision making because personalized management based on many variables, including patient trust, is essential for the success of any long-term treatment plan.25
Starting in 2013, the first biosimilars for the treatment of moderate to severe plaque psoriasis (etanercept, infliximab) and, by extrapolation, for other indications were introduced into the EU and their prescription was promoted by health care payers, who saw them as a way to reduce the cost of biologic therapy and possibly to achieve more equitable access to therapy in a number of health care systems.26 Their advent also led to a reduction in the price of the originator biologics and the introduction of reference prices for biologic therapies in Spain. However the economic impact of these changes was moderate and the impact on the cost of dermatological prescription was even less significant. This was due to the inconvenience of infliximab infusions and the limited effectiveness of etanercept compared to other therapies available for the treatment of psoriasis.
Following the expiration of the Humira® patent in the EU in 2018, the European Medicines Agency approved numerous adalimumab biosimilars (Amgevita®, Imraldi®, Cyltezo® [subsequently withdrawn], Hyrimoz®, Hulio®, Idacio®, and Amsparity®, to date). The introduction of these products quickly led to considerable price reductions, representing a discount of as much as 80% on the cost of adalimumab before the arrival of biosimilars. In Spain, even taking into account the reference price established for all the variants of adalimumab, market competition has led to offers that currently make this treatment as economical, or even less expensive, than ciclosporin and certain topical treatments, with the result that price no longer represents a significant barrier to its prescription.
According to a recently published network meta-analysis,27 the most effective biologic treatments belong to 2 groups: a) inhibitors that target interleukin (IL) 17A and its receptor; and b) IL-23(p19) inhibitors. TNF-α inhibitors are less effective (and are associated, in some cases, with a higher risk of infections and possibly nonmelanoma skin cancer in patients with psoriasis) although they are more effective than conventional systemic treatments. For example, the NNT (number of patients who need to be treated to achieve complete clearance in 1 patient) is less than 3 for ixekizumab, risankizumab, brodalumab and guselkumab, 4 for infliximab, close to 6 for adalimumab and guselkumab, almost 14 for etanercept, and around 50 for both dimethyl fumarate and apremilast. In a recent Cochrane review, the unit-rounded relative risk with respect to placebo of obtaining a PASI-90 response at the primary endpoints of clinical trials (8 to 24 weeks after randomization) was over 25 for infliximab, ixekizumab, risankizumab, and guselkumab, 24 for secukinumab, 22 for brodalumab, 18 for adalimumab, 17 for ustekinumab and tildrakizumab, 10 for ciclosporin, etanercept, and methotrexate, 7 for apremilast, 4 for fumaric acid, and 2 for acitretin.2
Thus, TNF-α inhibitor biosimilars (Table 1), and in particular adalimumab biosimilars, represent a first-line treatment that is safer and more effective in the long term than systemic treatments at a similar cost for patients with moderate to severe plaque psoriasis; they are also more efficient than even the newer biologic agents. Nevertheless, it should also be taken into account that a significant percentage of patients will present primary or secondary treatment failure or an adverse effect and will have to switch to another innovative active principle within a few months. This means that the initial financial advantage may, in the medium term, be undermined by the increased expense associated with a switch to an original biologic agents that costs significantly more. The expansion, due to these changes, in the prescription of both biosimilars and new biologic agents could occur in a scenario in which the authorities may have limited or reduced the budget allocated to biologic therapy in psoriasis based on their confidence in the savings achieved following the introduction of biosimilars. This could eventually result in a need to ration such therapy or to introduce waiting lists for its prescription.
In clinical practice, the time to discontinuation (drug survival) of biosimilars, at least in the treatment of psoriasis, appears to be similar to that of the reference drugs.28 However, it is difficult to study drug survival for biosimilars in real life when there is considerable pressure in favor of their initial prescription and also pressure to switch to or from a biosimilar product; moreover, findings based on the historical data are of doubtful validity owing to changes over time in prescription patterns.
Finally, the trend towards a higher rate of discontinuation following a switch to a biosimilar could be explained by the nocebo effect, that is, the patient’s subjective perception (unrelated to any objective characteristics of the drug) that the biosimilar will not be as safe or effective as the original biologic. This effect would be more evident in diseases in which assessment depends primarily on symptoms (such as rheumatological diseases or inflammatory bowel disease) but may be less applicable in others, for example the loss of response observed in some patients with psoriasis following a switch from originator biologic to biosimilar.9,29–31
In short, biosimilar drugs are subject to a specific regulation that, by incorporating structural analyses, pharmacokinetic studies and at least one clinical trial to compare them with the originator, guarantees their use in the same conditions as the reference drug. Since biosimilars must be similar in effectiveness and safety to the originator biologics, the benefits of their incorporation into health services and prescription are based exclusively on their lower cost and greater efficiency, advantages that improve as the acquisition price decreases.
The incorporation of biosimilars should facilitate access to biologic therapies for all the patients for whom they are indicated. Paradoxically, in a scenario that should improve our prescription possibilities, offering technical and economic efficiency, there has been an increase in intervention on the part of regulatory agencies (national, regional, and local), often focused exclusively on reducing costs. This type of intervention could limit—or may already have limited—the freedom of clinicians to make appropriate prescription decisions and may strain the relationship between physicians, hospital pharmacies, the regulatory agencies, and the authorities responsible for paying for health care (the regional health ministries).
In the opinion of the authors of this article, there is a need to align the objectives of all the parties involved with the common goal of offering the best treatment options to patients with moderate to severe psoriasis (which, by definition, requires systemic treatment).32 The choice of therapy should be based on criteria relating to efficiency rather than on criteria based solely on cost, which fail to take effectiveness into account. The criteria should not undermine the clinical objectives (reducing the impact of the disease to a minimum using the ideal treatment for each case) or prejudice the safety of the patient in each specific case. In many cases, these objectives will require first-line treatment with the available biosimilars (or innovative drugs that offer similar efficiency), leaving the second and subsequent lines of treatment to be decided by the specialist.
Conflicts of InterestL. Puig has received fees for acting as a consultant and/or speaker for and/or has participated in clinical trials sponsored by Abbvie, Almirall, Amgen, Baxalta, Biogen, Boehringer Ingelheim, Celgene, Gebro, Janssen, JS BIOCAD, Leo-Pharma, Lilly, Merck-Serono, MSD, Mylan, Novartis, Pfizer, Regeneron, Roche, Sandoz, Samsung-Bioepis, Sanofi, and UCB.
J.M. Carrascosa has received fees for acting as a consultant and/or speaker for and/or has participated in clinical trials sponsored by Abbvie, Almirall, Amgen, Biogen, Boehringer Ingelheim, Celgene, Gebro, Janssen, Leo-Pharma, Lilly, Mylan, Novartis, Pfizer, Amgen, Sandoz, Samsung-Bioepis, Sanofi, and UCB.
J. Notario has received fees for acting as consultant and/or speaker for and/or has participated in clinical trials sponsored by Abbvie, Almirall, Celgene, Gebro, Janssen, Leo-Pharma, Lilly, Merck-Serono, MSD, Novartis, Pfizer, Sandoz, and UCB.
Please cite this article as: Puig L, Carrascosa JM, Notario J. Biosimilares en el tratamiento de la psoriasis. Actualización. Actas Dermosifiliogr. 2020;111:809–814.