Atopic dermatitis (AD) is the most common inflammatory skin disease, affecting nearly 20% of the pediatric population, 15% of adolescents, and 10% of adults worldwide. AD is driven by a disruption of the skin barrier and dysfunction of T1 lymphocyte-dependent inflammatory pathways.1
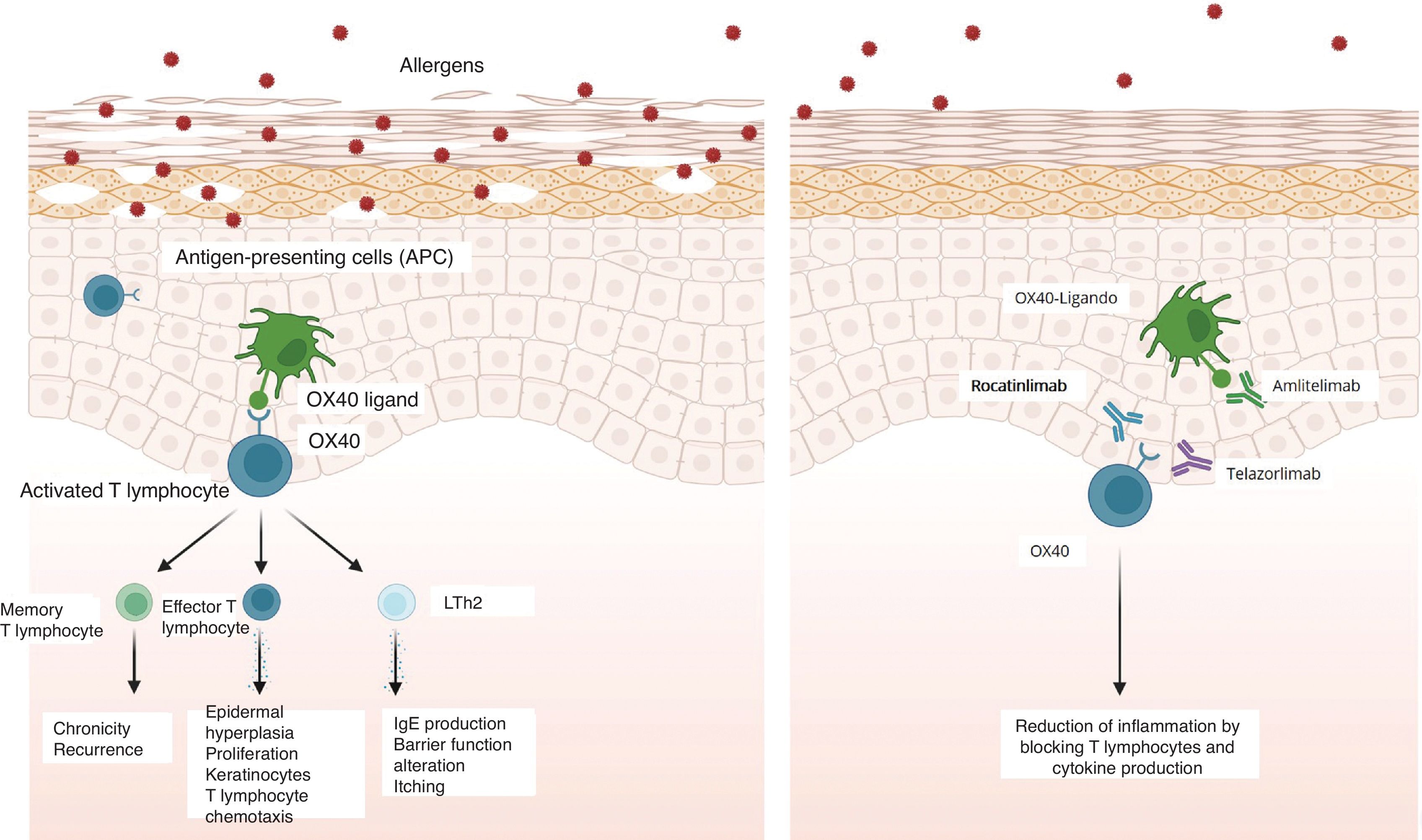
A member of the tumor necrosis factor receptor (TNFR) superfamily known as OX40 is a co-stimulatory molecule expressed on T lymphocytes between 24 and 72h after their activation. It binds to the OX40 ligand (OX40L; CD252) expressed on activated antigen-presenting cells at the site of inflammation.1 The OX40-OX40L complex enhances the proliferation, survival, adhesion, migration, cytokine production of effector T lymphocytes, Th2 lymphocytes, and promotes CD4+ memory T lymphocytes. In humans, OX40 expression has been observed specifically on T lymphocytes in the lesional skin of patients with immune-mediated disorders, including AD.2
Clinical trials have been conducted in moderate-to-severe AD (Fig. 1) with 3 OX40 antibodies (rocatinlimab [AMG 451/KHK4083-Amgen®, subcutaneous route] and telazorlimab [GBR 830]-Ichnos Sciences SA®, subcutaneous route) or OX40L (amlitelimab [KY1005]-Sanofi®, subcutaneous route).
OX40 inhibition model and therapeutic targets. Note: This figure was created by the authors with BioRender (Science Suite Inc®, Toronto, Canada) and is permitted for publication in scientific journals.
Source: Adapted from Croft et al.1.
Rocatinlimab3 is a non-fucosylated anti-OX40 monoclonal IgG1 antibody, which showed a significant reduction in EASI score in patients with moderate-to-severe AD. A phase 1 study revealed a 74% reduction in EASI from baseline up to week 22, with 35% of patients achieving IGA 0/1 on week 22. In a phase 2b trial, significant improvements were seen on week 16, with an 88% reduction in EASI on week 36. Biomarker modulation and tolerability support its potential as a durable and safe treatment for AD.
Telazorlimab (GBR 830/ISB 830)4 is a humanized IgG1 neutralizing monoclonal antibody against OX40 that blocks its interaction with OX40L, evaluated in phase 2a and 2b trials in AD patients. In the phase 2a trial (n=62), 77% of patients on telazorlimab showed a 50% improvement in EASI score on day 71, while 23% achieved IGA 0/1. In this trial, telazorlimab was well-tolerated, with headache being the most common adverse event. The phase 2b trial (n=462) showed a significant improvement in EASI with telazorlimab vs placebo. The most common adverse effects were nasopharyngitis, upper respiratory tract infection, and headache. Although no ongoing or planned trials for telazorlimab in AD are currently available, its development is likely to continue for rheumatoid arthritis or other autoimmune diseases.
Amlitelimab (KY1005)5 is a human IgG4 antibody that binds to OX40L and blocks its signaling. In the pivotal phase 2a study in moderate-to-severe AD patients (n=88), the low-dose regimen of amlitelimab (200mg loading/100mg maintenance every 4 weeks) showed an 80% improvement in the mean EASI score vs 49% in patients with placebo. The high-dose regimen (500mg loading/250mg maintenance every 4 weeks) resulted in a 70% improvement in EASI score. Headache, upper respiratory tract infections, and hyperhidrosis were the most common adverse effects.
The promising results of early-phase clinical trials with OX40 antibodies (rocatinlimab; telazorlimab) or OX40L (amlitelimab) in moderate-to-severe AD highlight the potential of targeting the OX40 pathway as a novel therapeutic approach for the treatment of AD.
FundingNone declared.
Conflicts of interestNone declared.
We wish to thank Álvaro Prados-Carmona, EIR Dermatology at HUSC, for his collaboration in the creation of the figure.